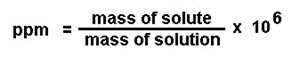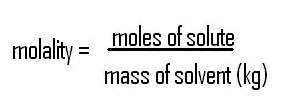Ways to measure concentration
There are several ways to express the concentration of a solution. The most common that we have used is molarity (M). But there is also % by mass, parts per million, molality, mole fraction, just to name a few. These are important to understand and how to convert from one unit to another because each one of the "colligitave properties" that we will study all rely on a different measurement of concnetration.
| CH 11 Study Guide Key- Full | |
| File Size: | 940 kb |
| File Type: | |
| CH 11 Study Guide | |
| File Size: | 667 kb |
| File Type: | |
| CH 11 SG Key p. 1-11 | |
| File Size: | 522 kb |
| File Type: | |
| Freezing Point Depression of PDCB Lab | |
| File Size: | 615 kb |
| File Type: | |