Homework for Monday, 12/2 (B Classes) and Tuesday, 12/3 (A Classes)
Complete up to page 8 in the mole packet.
THE MOLE
The term "mole" is a chemists' way of counting. I like to call it the "chemist's dozen." Similar to a dozen eggs being 12 eggs or a dozen elephants being 12 elephants, if you have a mole of atoms or molecules or pencils, you have 6.02 x 10^23 of them. A mole of iron has 6.02 x 10^23 atoms of iron and that many atoms of iron has a mass of 55.8 grams (the atomic mass of iron). Similarly, A mole of copper has 6.02 x 10^23 atoms of copper and that many atoms of copper has a mass of 63.5 grams (the atomic mass of copper).
Determining the Molar Mass
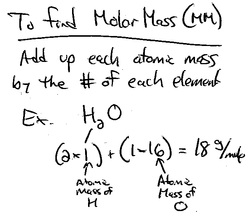
Click on the picture for a tutorial on calculating the molar mass of an element or a compound.
These are the steps in calculating the molar mass of a compound.
1. Count the # of atoms of each element in the compound.
2. Multiplay that number by the atomic mass of each element (from the periodic table)
3. Add up the masses of each element to get the total mass of one mole of a compound.
1. Count the # of atoms of each element in the compound.
2. Multiplay that number by the atomic mass of each element (from the periodic table)
3. Add up the masses of each element to get the total mass of one mole of a compound.
Using the Mole Finding Tool

Click on the image for a tutorial on mole calculations using the mole finding tool.
Using this "mole finding tool," you can easily convert masses of substances into moles and then into particles (atoms or molecules). You will need to know the molar mass of the compound.
To find % composition of elements in a compound
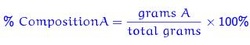
Click on the image for a tutoiral on calculating the percent composition of elements in a compound.
To find the percent composition of each element in a compound, you simply take the molar mass (unless specific masses are given) of the element in question and divide it by the total molar mass of the compound. That decimal is then multiplied by 100 to get the percentage. This percent is very important in determining the formula of a compound.
To find the emipirical (simplest) formula of a compound

Click on the image for a tutorial on determining the empirical formula of a compound.
The empirical formula is the simplest, whole # ratio of elements in a compound. In order to find the ratio, you need to convert the mass (in grams) or the % of each compound into moles. You can use the "formula finding tool" to calculate the empirical formula.
To find the molecular formula of a compound.
The molecular formula is a whole # multiple of the ratio in the empirical formula.
The steps to find the molecular formula of the compound are as follows.
1. Calculate the empirical formula of the compound (see above)
2. Calculate the mass of one mole of the empirical formula (empirical mass)
3. Divide the molar mass of compound (given in the problem) by the mass of the empirical formula (step 2). This answer should be a whole #. If it isn't, you either incorrectly found the empirical formula or the mass of the empirical formula.
4. Multiply your ratio (empirical form) by the whole # (step 3). This new ratio is the ratio of elements in the compound of the molecular formula.
If you are unsure of any of these steps, you can refer to the tutorial here.
The steps to find the molecular formula of the compound are as follows.
1. Calculate the empirical formula of the compound (see above)
2. Calculate the mass of one mole of the empirical formula (empirical mass)
3. Divide the molar mass of compound (given in the problem) by the mass of the empirical formula (step 2). This answer should be a whole #. If it isn't, you either incorrectly found the empirical formula or the mass of the empirical formula.
4. Multiply your ratio (empirical form) by the whole # (step 3). This new ratio is the ratio of elements in the compound of the molecular formula.
If you are unsure of any of these steps, you can refer to the tutorial here.
| Mole Packet of Problems | |
| File Size: | 395 kb |
| File Type: | |
| Mole Packet Key | |
| File Size: | 753 kb |
| File Type: | |
| Tutorial Worksheet on determining molar masses | |
| File Size: | 142 kb |
| File Type: | |
| Iron-Copper Sulfate Lab Key | |
| File Size: | 91 kb |
| File Type: | |
| Copper-Silver Nitrate Lab Key | |
| File Size: | 108 kb |
| File Type: | |
