Graham's Law of Effusion & Diffusion
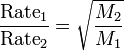
This basically says that a heavier gas will travel slower through the air than a lighter gas. It explains why pleasant (or unpleasant) odors seem to linger and then disappear. The gas molecules are in constant motion and it takes the odor with it. Click here for a tutorial on Graham's Law.
Gaseous Stoichiometry

This is the Barry Bonds version of the Mole Finding Tool.
It's back again- stoichiometry! No worries, it is a little easier this time around. If you are comparing gases that are under identical pressure and temperature conditions, you can use the volume of gases similarly to moles of a gas, the balanced equation gives you both mole ratios AND volume ratios.
However, you can also use the ideal gas law (PV = nRT) to determine moles of gases. You would then use your balanced equation to get mole ratios and then continue to use stoichiometry.
You can also use the molar volume of a gas (22.4 L/mole) to find out the number of moles of gas at STP. This only works under standard temperature and pressure conditions (1 atm & 273 K). Click here for a tutorial.
There are some detailed notes that might help you with gaseous stoichiometry.
However, you can also use the ideal gas law (PV = nRT) to determine moles of gases. You would then use your balanced equation to get mole ratios and then continue to use stoichiometry.
You can also use the molar volume of a gas (22.4 L/mole) to find out the number of moles of gas at STP. This only works under standard temperature and pressure conditions (1 atm & 273 K). Click here for a tutorial.
There are some detailed notes that might help you with gaseous stoichiometry.
| Full Key for Gas Packet | |
| File Size: | 743 kb |
| File Type: | |
