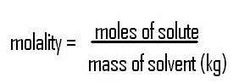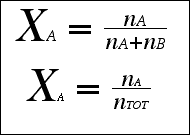|
Here is a pdf file of your packet on solutions, as well as the answer key to the first 11 pages of the packet, which include the differences between solutions, suspensions & colloids as well as the parts of solutions and the answers to the molarity problems we worked on in class or for homework.
|
|
| ||||||||||||||||||||||||||||||||||||
Dilutions
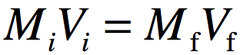
When you have a solution that is too strong of a concentration, you may wish to dilute it or water it down. A dilution is when you add solvent, usually water, to a solution. This will make the volume of solution go up, but the moles of solute remains the same. This results in a lowering of the Molarity of the solution. The moles before the dilution will equal the moles after the dilution. From the molarity formula, moles = M x V, so moles 1 = moles 2, so M1V1 = M2V2.
Solubility Curves

This is a common solubility curve. Notice the 2 compounds that have decreasing lines, they are gases.
One way to tell how saturated or unsaturated or supersaturated a solution is to refer to a solubility table. This table will tell you how many grams of a solute will dissolve in 100 g of water at a given temperature. For most substances (solids), the solubility increases with temperature. That is, you will be able to dissolve more solute in 100 g of water as the temperature increases. The rare exceptions are gases; the solubility of gases decreases with an increase in temperature. This is the reason room temperature soda fizzes more than soda kept in the fridge. The warmer room air will not allow as much carbon dioxide to be dissolved in the soda, so it fizzes out.
When you use the curve, it is a starting point. Sometimes you want to dissolve solute in more than 100 g of water, sometimes you will need less. You find the ratio of g solute/100 g water at a given temperature and set up a ratio to figure out the proper amount of solute or the proper amount of water.
Any point above the line is considered supersaturated. (It contains more than the maximum amount of solute dissolved.) Any point below the line is considered unsaturated. (It contains less than the maximum amount of solute. Any point on the line is considered saturated. (It contains the maximum amount of solute the solution can hold.)
When you use the curve, it is a starting point. Sometimes you want to dissolve solute in more than 100 g of water, sometimes you will need less. You find the ratio of g solute/100 g water at a given temperature and set up a ratio to figure out the proper amount of solute or the proper amount of water.
Any point above the line is considered supersaturated. (It contains more than the maximum amount of solute dissolved.) Any point below the line is considered unsaturated. (It contains less than the maximum amount of solute. Any point on the line is considered saturated. (It contains the maximum amount of solute the solution can hold.)