Acids & Bases
Acids are hydrogen donors. They are sour and have a pH of less than 7. When a metal (magnesium or zinc)is added to an acid, hydrogen gas is produced. When a carbonate (like TUMS) is added to an acid, carbon dioxide is produced. If blue litmus paper is used in testing acids, it will turn red. Red litmus paper will not change color with an acid. Acids appear clear when phenolphthalein is added to it. Acids are red when universal indicator is used.
Bases are hydrogen acceptors. They are bitter and have a pH of greater than 7. Bases do not react with metals like magnesium or zinc. Bases also do not react with carbonates. If red litmus paper is used in testing bases, it will turn blue. Blue litmus paper will not change color with a base. Basess appear pink when phenolphthalein is added to it. Bases are purple when universal indicator is used.
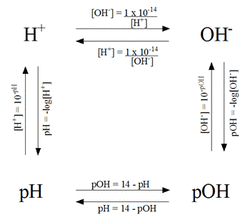
The pH scale is used to measure the acidity or basicity of a substance. pH is calculated using this formula: pH = -log[H+]. pOH is calculated using this formula: pOH = -log[OH-]. The sum of pH and pOH of a substance is always 14.
A neutralization reaction occurs when an acid is titrated (added) to a base or vice versa. The acid and bases neutralize or cancel each other out. The general formula is: Acid + Base --> Salt + Water. You can calculate the molarity of an acid or base by using the dilution formula (MaVa = MbVb).
Bases are hydrogen acceptors. They are bitter and have a pH of greater than 7. Bases do not react with metals like magnesium or zinc. Bases also do not react with carbonates. If red litmus paper is used in testing bases, it will turn blue. Blue litmus paper will not change color with a base. Basess appear pink when phenolphthalein is added to it. Bases are purple when universal indicator is used.
The pH scale is used to measure the acidity or basicity of a substance. pH is calculated using this formula: pH = -log[H+]. pOH is calculated using this formula: pOH = -log[OH-]. The sum of pH and pOH of a substance is always 14.
A neutralization reaction occurs when an acid is titrated (added) to a base or vice versa. The acid and bases neutralize or cancel each other out. The general formula is: Acid + Base --> Salt + Water. You can calculate the molarity of an acid or base by using the dilution formula (MaVa = MbVb).
Oxidation-Reduction Reactions

An oxidation-reduction reaction (RedOx) involves a transfer of electrons. If a substance gains electrons, it is said to be reduced. If a substance loses electrons, it is said to be oxidized. The simple acronym to remember this is OIL RIG (Oxidation Is Loss (electrons) Reduction Is Gain (electrons)).
To tell if something is being oxidized or reduced, you need to check out its oxidation #. If a substance's oxidation # goes up from one side of a reaction to another, it is being oxidized (losing electrons, resulting in an increased oxidation #). If a substance's oxidation # goes down from one side of a reaction to another, it is being reduced (gaining electrons, resulting in a decreased oxidation #).
Here are the rules for identifying oxidation #'s for elements or ions.
To tell if something is being oxidized or reduced, you need to check out its oxidation #. If a substance's oxidation # goes up from one side of a reaction to another, it is being oxidized (losing electrons, resulting in an increased oxidation #). If a substance's oxidation # goes down from one side of a reaction to another, it is being reduced (gaining electrons, resulting in a decreased oxidation #).
Here are the rules for identifying oxidation #'s for elements or ions.
- The oxidation number of a free, uncombined element (including elements in the molecular state) is zero.
- Oxygen always has an oxidation number of –2 in any compound except in peroxides where it is –1.
- Hydrogen has an oxidation number of +1 except when combined with a metal in a simple binary compound where it is –1.
- The algebraic sum of all valences and/or oxidation numbers in any compound must equal zero.
- The algebraic sum of all the valences and/or oxidation numbers in a polyatomic ion must add up to the charge carried by that ion.
|
| ||||||||||||
