A Chemical Reaction
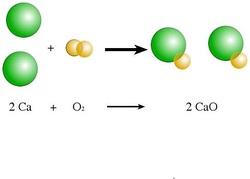
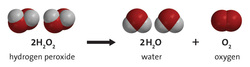
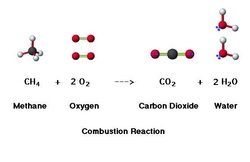
Any reaction can be written as a balanced chemical equation. This is like the recipe of what is mixing. Every reaction follows the format: reactants ---> products. This translates into "reactants change into products." As in any chemical reaction, the law of conservation of matter must be obeyed. This means that the # of atoms on each side of the arrow must be the same. If they aren't, you can "balance" them by adding a coefficient in front of the element or compound. You will do this until all elements are balanced.
5 Reaction Types
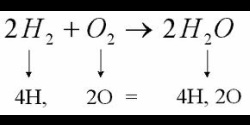
Click on the picture for a 5 minute tutorial video on balancing equations.
There are 5 types of reactions will we use most often in this class. They are synthesis, decomposition, single replacement, double replacement and combustion reactions. They each follow a certain pattern
CH 11- Stoichiometry
Stoichiometry is simply just a chemists mathematical way of following a recipe, or a balanced equation. The balanced equation gives you the ratios of MOLES that each reactant and product are present in the reaction. In actuality, one rarely uses full moles when working in the lab. The amounts you use can be converted into moles and then the mole ratios from the balanced equation can be used.
Here are the 4 basic steps to any stoichiometric problem (these are the basics and they can get more complicated, but it still follows the same rules.)
1. Balance an equation (you can't do anything in stoichiometry unless you have the correct mole ratios)
2. Convert the information given into moles (I will call this "moles given" throughout these steps.)
3. Using the mole ratios in the balanced equation, convert the moles given into moles of what the question is asking. (I call this "moles needed."
4. Convert the moles needed into whatever the question is asking for (grams, liters, milliliters, etc).
Here are the 4 basic steps to any stoichiometric problem (these are the basics and they can get more complicated, but it still follows the same rules.)
1. Balance an equation (you can't do anything in stoichiometry unless you have the correct mole ratios)
2. Convert the information given into moles (I will call this "moles given" throughout these steps.)
3. Using the mole ratios in the balanced equation, convert the moles given into moles of what the question is asking. (I call this "moles needed."
4. Convert the moles needed into whatever the question is asking for (grams, liters, milliliters, etc).
Types of stoichiometric Calculations
Mole-Mole Calculations
Click on the link here to show a 3 minute video clip on mole to mole stoichiometric calculations.
Gram to Gram calculations.
Click on the link here to show a 5 mintue video clip on gram to gram stoichiometric calculations.
Click on the link here to show a 3 minute video clip on mole to mole stoichiometric calculations.
Gram to Gram calculations.
Click on the link here to show a 5 mintue video clip on gram to gram stoichiometric calculations.
Limiting Reactant Problems
A limiting reactant problem occurs when you are given two or more reactants and you need to find out how many grams of a certain product will form. If the reactant amounts are not in exact mole ratios, this problem would become tricky. But, all is not lost. What you need to do is find out which reactant would produce the LEAST amount of product. The reactant that produces the least product will be the "limiting reactant," that is is "limits" the amount of product to be formed. There are several ways to look at these problems, I like to look at them to see which one produces the least amount of product. Click here for a tutorial video on limiting reactants.
% Yield
When you do a reaction on paper, you will go through the stoichiometry and you will get what is called a "theoretical yield" or answer. This is how much you "should" form, if all goes well in the lab. Well, as you know, not everything goes as planned in the lab. There are dozens of errors that could alter your answer. If you do the lab, your product is called an "actual yield." Your "grade" as to how well the reaction went is in the form of a % yield, or what percent of what you should have gotten did you really get. The formula is simple:
% yield = actual yield x 100
theoretical yield
% yield = actual yield x 100
theoretical yield
| Packet Key All pages | |
| File Size: | 932 kb |
| File Type: | |
| Chalk Lab Key | |
| File Size: | 182 kb |
| File Type: | |