| CH 10 Study Guide | |
| File Size: | 858 kb |
| File Type: | |
| Chapter 10 Study Guide Key | |
| File Size: | 1140 kb |
| File Type: | |
10.1 Intermolecular Forces
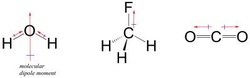
Three intermolecular forces- Dipole-dipole (force of attraction between the positive dipole of one molecule with the negative dipole of another molecule). Not very strong.
Hydrogen Bonding- Dipole interaction where Hydrogen is at one end. VERY STRONG
London Dispersion Forces- forces between noble gases or between nonpolar molecules. Very weak.
Hydrogen Bonding- Dipole interaction where Hydrogen is at one end. VERY STRONG
London Dispersion Forces- forces between noble gases or between nonpolar molecules. Very weak.
10.2 Vapor Pressure and Changes of State.
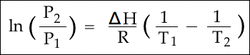
This section deals with the Clausius-Clapeyron Equation which equates the vapor pressure of a liquid at a certain temperature with the vapor pressure of a liquid at a different temperature.
Warming Curves
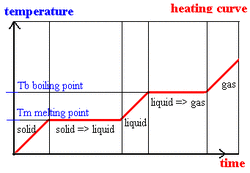
This warming curve can show how much heat is required to melt ice (molar heat of fusion), how much heat is required to warm water (Q = mct) or to vaporize water (molar heat of vaporization).
Phase Diagrams
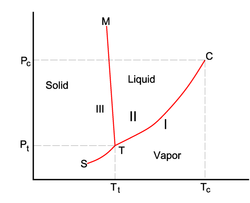
This diagram shows what happens to the state of matter of a substance as temperature or pressure is change. The point marked "T" is the triple point, which is the temperature and pressure where the solid and liquid phase have the same vapor pressure. The point marked "C" is the critical point, which is the point on the diagram where the critical temperature and the critical pressure meet. The critical temperature is the temperature when a vapor can't be liquified regardless of the pressure. The critical pressure is the pressure required to produce liquification at the critical temperature.
If the solid/liquid line has a negative slope, the solid is less dense than the liquid (water is an example). If the solid/liquid line has a positive slope, the solid is MORE dense than the liquid (carbon dioxide is an example).
If the solid/liquid line has a negative slope, the solid is less dense than the liquid (water is an example). If the solid/liquid line has a positive slope, the solid is MORE dense than the liquid (carbon dioxide is an example).
Liquid-Vapor Equilibrium

If a liquid is left in an open container, it will evaporate completely. However, if the container is closed, the vapor will have no place to escape, so it will condense into a liquid again. Eventually, the rate of vaporization = rate of condensing. In any kind of vapor equilibrium problem, you can treat the vapor like an ideal gas and finish the calculations using the Ideal Gas Law. (PV = nRT).
